Artificial Nitrogen Fertilizer-Dangers Of Mankind Playing God
There should be a law against mankind playing God over nature. Human beings are superior but they are also greedy. Never satisfied with what nature has to offer. We want more and because we are superior we find ways to imitate nature and trick her into doing what we want.
We make decisions and never stop to ask ourselves what the consequences of these decisions will be. A classical example of mankind playing God is the introduction of synthetic fertilizer in the form of artificial nitrogen just over a century ago. Nature could not provide us with a big enough crop yield so we went and found a way of imitating what nature does and forced her to give us bigger crops. Now 100 years later we wake up to the realization that by doing so we have poisoned our environment.
What is Nitrogen?
The Earth's atmosphere consists of 70-80% nitrogen. It is essential to all life and is a crucial element of food production. Most biological processes are dependent on nitrogen as it is an essential ingredient needed to build amino acids which form proteins.
Nitrogen in its natural state is mainly unusable by plants and needs to be converted from gaseous nitrogen into reactive nitrogen. The conversion of gaseous nitrogen to reactive nitrogen is known as fixation. Reactive nitrogen is a vital nutrient in soil and its abundance or scarcity will determine the fertility of the soil.
Nitrogen cycle
The nitrogen cycle is the continuous flow of nitrogen through the biosphere by the processes of nitrogen fixation, ammonification (decay), nitrification and denitrification.
The Nitrogen Cycle
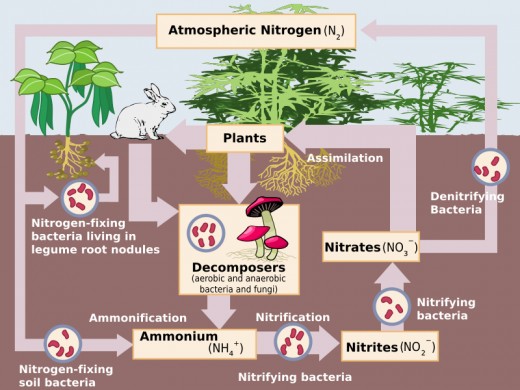
Nitrogen fixation
In order for nitrogen to be absorbed by the soil nitrogen must be “fixed” by adding hydrogen or oxygen which converts the nitrogen into compounds such as ammonia or nitrates that plants can utilize. Nitrogen-fixing bacteria are the only micro-organisms that are capable of absorbing nitrogen from the air and transforming it into nitrates. These are then absorbed by plants to build essential proteins.
A certain amount of atmospheric nitrogen is fixed by lightning and by some blue-green algae (cyanobacteria). The great bulk of nitrogen fixation however, is performed by soil bacteria. The are two kinds of soil bacteria. Those that live freely in the soil use the energy from decaying organic matter in the soil to stimulate soil processes including nitrogen fixation. The other type of soil bacteria, known as rhizobia, live in the nodules of leguminous plants such as Lucerne, peas, beans, clover, soybeans, and peanuts. When animals consume these plants the plant protein is converted into animal protein.
Nitrifying bacteria
When an organism produces waste or dies, the decomposing matter returns organic nitrogen to the soil as ammonia. Nitrifying bacteria then oxidize the ammonia into nitrites and then into nitrates, a process known as nitrification. The nitrates can then be absorbed from the soil water by the roots of the plant and used to make proteins.
Denitrification occurs in the absence of oxygen in the soil and is not common in well-cultivated soils. During this process a third group of bacteria turn nitrates back into nitrites and ammonia and sometimes even back into nitrogen gas. This is harmful to plants as they need nitrates to make protein.
The Introduction of Artificial Nitrogen
Soils recycle nitrogen for re-use in organic waste such as animal dung. In the past some countries improved the fertility of their soils by adding guano and saltpetre, which are sources of geological nitrogen. Until 1908 the only way of adding more atmospheric nitrogen to soils was through capture by the bacteria that live in a small number of nitrogen-fixing plants such as clover and beans.
In an effort to increase the fertility of soil an appeal was made to scientists to find a way of producing nitrogen in a form that plants could absorb. In 1908 Fritz Haber invented a cheap new source of nitrogen fertilizer when he discovered how to make ammonia, a molecule made of hydrogen and nitrogen atoms from the inert nitrogen gas in the air.
This was welcomed throughout the world and Haber received the 1918 Nobel Prize in chemistry for his work. Unfortunately nobody thought of the consequences of adding massive amounts of artificially-made nitrogen into the planet’s ecosystems.

Any High School chemistry student will recall having heard about the Haber Process for the synthesis of ammonia (NH3) gas from its elements nitrogen (N2) and hydrogen (H2) as this is often referred to as one of the best examples of chemical equilibrium. The Chemistry texts however very rarely give any indication of the effects that the discovery of this process has had on history or society.
The Path Of Destruction
The benefits that artificial nitrogen have of boosting the quantity of crops can not be denied but this has come at a price as far as quality is concerned. Artificial nitrogen fertilizers increase the level of potentially toxic nitrates, and other non-protein nitrogenous compounds as well as the sugars in the crops while decreasing the fibre content in the plants. Artificial nitrogen also impoverishes the soil by lowering the levels of trace minerals in the ground. The result is that these valuable nutrients are also substantially less in these crops.
These fertilizers also increase nitrates in our drinking water and damage vital soil micro flora. It reduces plant diversity in pasture land and contributes to reducing the quality of habitat for many insects. As a result birds that depend on these species of insects can not survive and need to migrate to other areas. It also influences the quality of the food we eat and is not the ideal for our fruit, vegetables and cereals.
The production of artificial nitrogen is now being linked to the dramatic spikes in population growth, energy use, and carbon production during the last half of the 20th century. All species flourish and multiply when food is abundant – mankind is no different. The introduction of artificial nitrogen fertilizers helped the human population multiply from 1.9 billion in 1900 to nearly 6.87 billion in 2008.
Dead Zone California

Nitrogen is to the Earth's Water as Carbon Dioxide is to its Air
Each year an average of 80 million tons of Haber’s fertilizer is spread onto fields around the world. Only 17million tons actually goes into food and the rest gets washed into the ecosystems.
Until recently people clearly refused to acknowledge the long-term damage that the use of artificial nitrogen is doing to water bodies, ecosystems, agricultural lands and human health. The observed increases in short-term crop yields derived from using artificial nitrogen can not make up for the long-term damage. We also fail to consider the carbon footprint and massive energy inputs of agrochemicals.
Artificial nitrogen has become as much a pollutant of the Earth’s water as carbon dioxide is to the air. It is accumulating in rivers around the world and is present in underground water reserves. This over-fertilization of the water produces large volumes of algae which consume all the oxygen present in the water, causing ecosystems to crash.
On land, nitrogen over-saturation has disrupted soil chemistry and created a depletion of other critical nutrients including calcium, magnesium, and potassium. Ironically, the addition of one nutrient out of balance with others can result in an overall decline in soil fertility, leading to diminished productivity of both cultivated and natural landscapes.
Nitrogen Pollution
Crossing the Planetary Boundary
Denitrification is nature’s way of reversing man-made fixing of nitrogen by converting it back to an inert gas but we have crossed the boundary and many of the rivers have become so nitrogen-saturated that they are losing their ability to denitrify pollution. The excess nitrogen ends up in the ocean causing oxygen-depleted “dead zones” where it is killing entire ecosystems.
According to a report by Johann Rockstrom and 27 scientists a safe boundary of nitrogen in the environment would be 35 million tons of nitrogen. Human activity currently releases 121 million tons of nitrogen into the environment creating serious distortions in Earth’s natural nitrogen cycle. Most of this ends up in the Earth’s waterways resulting in irreversible damage and loss of ecosystems. Based on current trends, global nitrogen use on farmland is set to double to 220 million tons a year by 2050 – more than six times the safe threshold.
The danger is that nature’s ability to process this excess nitrogen and return it to the atmosphere will be overwhelmed, and we will end up inhabiting a nitrogen-saturated planet, with nitrogen killing the oceans, driving global warming, acidifying air, depleting the ozone layer and reducing biodiversity.
Other Boundaries That Mankind Has Crossed
- Environmental Abuse - Biting The Hand That Feeds Us
Environmental abuse is inexcusable - it is the proverbial biting the hand that feeds us. Abuse occurs when the abuser violates the personal boundaries of their victims. The motivation for doing so is to...



